
Chemistry, 12.07.2019 18:00 Uniquestudies
Below is a proposed two-step mechanism for the decomposition of hydrogen peroxide: h2o2(aq) + br−(aq) → h2o(ℓ) + bro−(aq) slow bro−(aq) + h2o2(aq) → h2o(ℓ) + o2(g) + br−(aq) fast is this homogeneous or heterogeneous catalysis? also identify the catalyst in the mechanism.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:00
Which statement describes covalent bases? they have hydroxide ions. they produce hydrogen ions. they are often amines. they are named the same as ionic compounds.
Answers: 3

Chemistry, 22.06.2019 18:20
Categorize them by metal, nonmetal, in periodic tableductilenon-ductilemalleableoften gain electrons easilygood conductorpoor conductorcan be liquidselements
Answers: 2

Chemistry, 23.06.2019 02:10
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2

Chemistry, 23.06.2019 09:30
What is the force of an object when it landed(sitting in the ground)
Answers: 2
You know the right answer?
Below is a proposed two-step mechanism for the decomposition of hydrogen peroxide: h2o2(aq) + br−(a...
Questions



Spanish, 14.04.2020 19:23


Mathematics, 14.04.2020 19:23

Mathematics, 14.04.2020 19:23








Mathematics, 14.04.2020 19:23



Mathematics, 14.04.2020 19:23


History, 14.04.2020 19:24

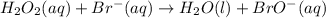 .....Slow
.....Slow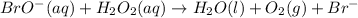 .....Fast
.....Fast

