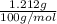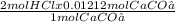Chemistry, 14.07.2019 00:00 ayoismeisalex
You measured out 1.212g caco3 to create the calcium supplement. how many ml of 1m hcl would you need to completely dissolve the caco3?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Iknow the answer to 13 is b and 14 is d. i just need to know why the correct answers are correct
Answers: 1

Chemistry, 21.06.2019 22:30
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

You know the right answer?
You measured out 1.212g caco3 to create the calcium supplement. how many ml of 1m hcl would you need...
Questions

History, 11.09.2021 14:00

Physics, 11.09.2021 14:00

Mathematics, 11.09.2021 14:00


Health, 11.09.2021 14:00





Mathematics, 11.09.2021 14:00

Mathematics, 11.09.2021 14:00





Mathematics, 11.09.2021 14:00

History, 11.09.2021 14:00


English, 11.09.2021 14:00

English, 11.09.2021 14:00