Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 23.06.2019 07:00
Write a hypothesis that answers the lesson question, “while observing a chemical reaction, how can you tell which reactant is limiting? ” hypothesis: if a substance is the limiting reactant, then . . because . .
Answers: 1

Chemistry, 23.06.2019 07:20
Which statement explains which component is likely to be more powerful in explaining a scientific phenomenon? a) component c, because a theory is often passed on possibility and not certainty b) component d, because a hypothesis is often based on possibility not certainty c) component c, because the ability to explain several occurrences in the natural world is a characteristic of a hypothesis d) component d, because the ability to explain several occurrences in the natural world is a characteristic of a theory
Answers: 3

You know the right answer?
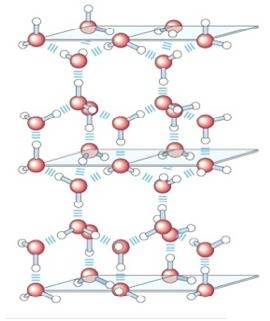
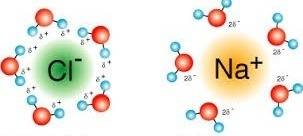
How is it that when a salt sample dissolves in water, the delta s for the process is positive?...
Questions

History, 18.04.2020 02:40

Chemistry, 18.04.2020 02:40


Social Studies, 18.04.2020 02:40


Chemistry, 18.04.2020 02:40

Mathematics, 18.04.2020 02:40


Mathematics, 18.04.2020 02:40

Mathematics, 18.04.2020 02:40


Computers and Technology, 18.04.2020 02:40



Computers and Technology, 18.04.2020 02:40

Mathematics, 18.04.2020 02:40

Mathematics, 18.04.2020 02:40