
Chemistry, 15.07.2019 12:00 live4dramaoy0yf9
Check all that apply to kcl. dissociates in water is an electrolyte is a nonelectrolyte is a salt

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 22:00
Ill give u brainliest pls how is mass of carbon conserved during cellular respiration
Answers: 1

Chemistry, 23.06.2019 06:00
Each step in the following process has a yield of 70% ch4 + 4cl2 yield ccl4 +4hcl ccl4 + 2hf yield ccl2f2 + 2hcl of 4.50 mole ch4 reacts what is the total amount of hcl produced
Answers: 3

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
Check all that apply to kcl. dissociates in water is an electrolyte is a nonelectrolyte is a salt...
Questions

Mathematics, 18.05.2021 01:00


English, 18.05.2021 01:00


SAT, 18.05.2021 01:00

Engineering, 18.05.2021 01:00

Mathematics, 18.05.2021 01:00


Mathematics, 18.05.2021 01:00








Mathematics, 18.05.2021 01:00


History, 18.05.2021 01:00

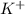 )and a non metal (
)and a non metal (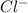 ).
). 


