Answers: 2


Another question on Biology


Biology, 22.06.2019 08:40
What best explains whether bromine (br) or neon (ne) is more likely to form a covalent bond? bromine forms covalent bonds because it has seven valence electrons, but neon has eight valence electrons and already fulfills the octet rule. bromine forms covalent bonds because it has many electron shells, but neon has only two electron shells and is tightly bound to its electrons. neon forms covalent bonds because it can share its valence electrons, but bromine has seven valence electrons and can gain only one more electron. neon forms covalent bonds because it has only two electron shells, but bromine has many electron shells and will lose electrons in order to fulfill the octet rule.
Answers: 3


Biology, 22.06.2019 22:30
Why can cancer in the lymph nodes (lymphoma) be particularly dangerous? a. the lymph node's ability to produce cancer-fighting lymphocytes would be compromised. b. the lymph node's ability to produce cancer-fighting lymph would be compromised. c. the lymph nodes produce lymph, which nourishes growing cancer cells. d. the lymph nodes produce chyle, which nourishes growing cancer cells.
Answers: 1
You know the right answer?
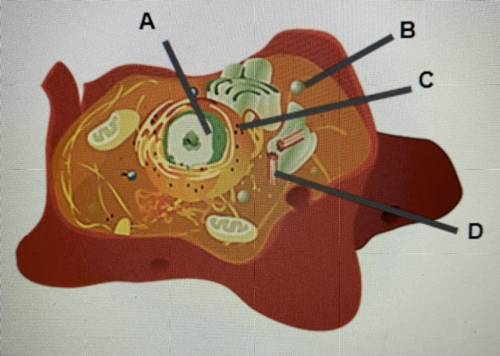
Identify organelles in an animal cell.
Answers;
Label A- Nucleus
Label B-...
Label A- Nucleus
Label B-...
Questions

English, 20.10.2021 07:30



Mathematics, 20.10.2021 07:30

Mathematics, 20.10.2021 07:30



Social Studies, 20.10.2021 07:30





Biology, 20.10.2021 07:30


Physics, 20.10.2021 07:30

English, 20.10.2021 07:30


English, 20.10.2021 07:30


Mathematics, 20.10.2021 07:30