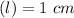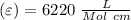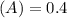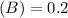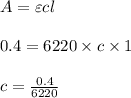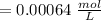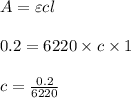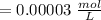Using and spectrophotometer, you measure 2 dilutions of NADH, and get absorbance values of 0.4 for sample A, and 0.2 for sample B. You know that the path length is 1 cm, and the extinction coefficient for NADH is 6220 (L Morcm). Using the Lambert-Beer Law equation (below), calculate the concentrations of sample A Select] and Sample B (Select ] A = log10 () = Ecl Where: A- Absorbance C- Concentration (mol 1 - Path length (cm) E = molar decadic extinction coefficient L mol. cm 1o - Intensity of the incident light 1 - Intensity of the transmitted Night

Answers: 3


Another question on Biology

Biology, 22.06.2019 07:00
Give two examples of what would occur if one of the pairs did not match or had an extra chromosomes. explain how this might occur, and if it would be dangerous to the individual. fast.
Answers: 1

Biology, 22.06.2019 11:00
1. which of the following transport mechanisms utilizes energy? a. osmosis b. diffusion c. facilitated diffusion d. endocytosis
Answers: 1

Biology, 22.06.2019 16:40
During dna replication,each strand of dna is used as a template to produce a complementary strand of dna. this process is shown below. which base will attach to location
Answers: 3

Biology, 22.06.2019 17:30
Select the correct answer. what does the hardy-weinberg principle relate to? a. chances of survival of an organism b. frequency of alleles in a population c. natural selection in a species d. causes of evolution among organisms
Answers: 2
You know the right answer?
Using and spectrophotometer, you measure 2 dilutions of NADH, and get absorbance values of 0.4 for s...
Questions

History, 26.01.2021 22:10




Business, 26.01.2021 22:10



Mathematics, 26.01.2021 22:10

Mathematics, 26.01.2021 22:10

Mathematics, 26.01.2021 22:10


Advanced Placement (AP), 26.01.2021 22:10

Mathematics, 26.01.2021 22:10

Chemistry, 26.01.2021 22:10

Health, 26.01.2021 22:10


Mathematics, 26.01.2021 22:10

Computers and Technology, 26.01.2021 22:10

Social Studies, 26.01.2021 22:10

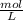 and concentration of sample B is 0.00003
and concentration of sample B is 0.00003