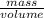1. A piece of pure gold (Au) has a volume of 50 cmWhat is its mass?
Density of Some Metals
Me...


Answers: 1


Another question on Biology

Biology, 22.06.2019 02:50
Cells destroy body cells infected by a virus or bacteria. killer t t memory t none of the above
Answers: 2



You know the right answer?
Questions




Biology, 02.11.2020 17:20

Mathematics, 02.11.2020 17:20



History, 02.11.2020 17:20










Biology, 02.11.2020 17:30

History, 02.11.2020 17:30

History, 02.11.2020 17:30